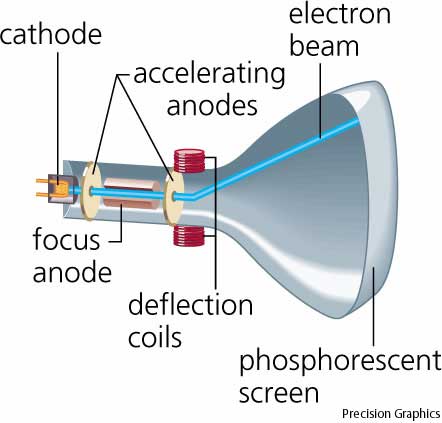
(See object ID EM.N-08253, Geissler tube demonstration apparatus.) In the early 1890s attention focused upon the rays which, at the highest evacuations, stream out from the negative pole (cathode) of the discharge tube and produce a fluorescent glow of its glass walls. The development of theory and experimental technique raised expectations of insightfrom this quarter into the structure of the atom and the connection between matter and electricity. 
History In the second half of the nineteenth century the phenomena accompanying the discharge of electricity through highly rarified gases received increasing attention from physicists. Thomson - the Centenary of His Discovery of the Electron and his invention of Mass Spectrometry, Rapid Communications in Mass Spectrometry, Vol.11, 2-16 (1997). Thomson, the Cavendish Laboratory, and Thompson's cathode ray tube and positive ray apparatus, see J. Used to determine the ratio of charge to mass (q/m) of the electron. External magnetic field can be applied to balance electric deflection. Metal plates can have electric field applied across them. (A separate image of the damaged object EM.N-08013-B will be added to this record in the near future.) Basic principle Rays from cathode in cylinder pass through 2 metal slots and between 2 metal plates onto glass ball. In its place, an identical object EM.318205, from a separate Museum collection (Electricity), can be seen 2nd from the left in the accompanying image. The object EM.N-08013-B was damaged after it was removed from the Museum's Atom Smashers exhibition. Five shorter glass tubes with internal wires extend perpendicularly from the cylinderical body. 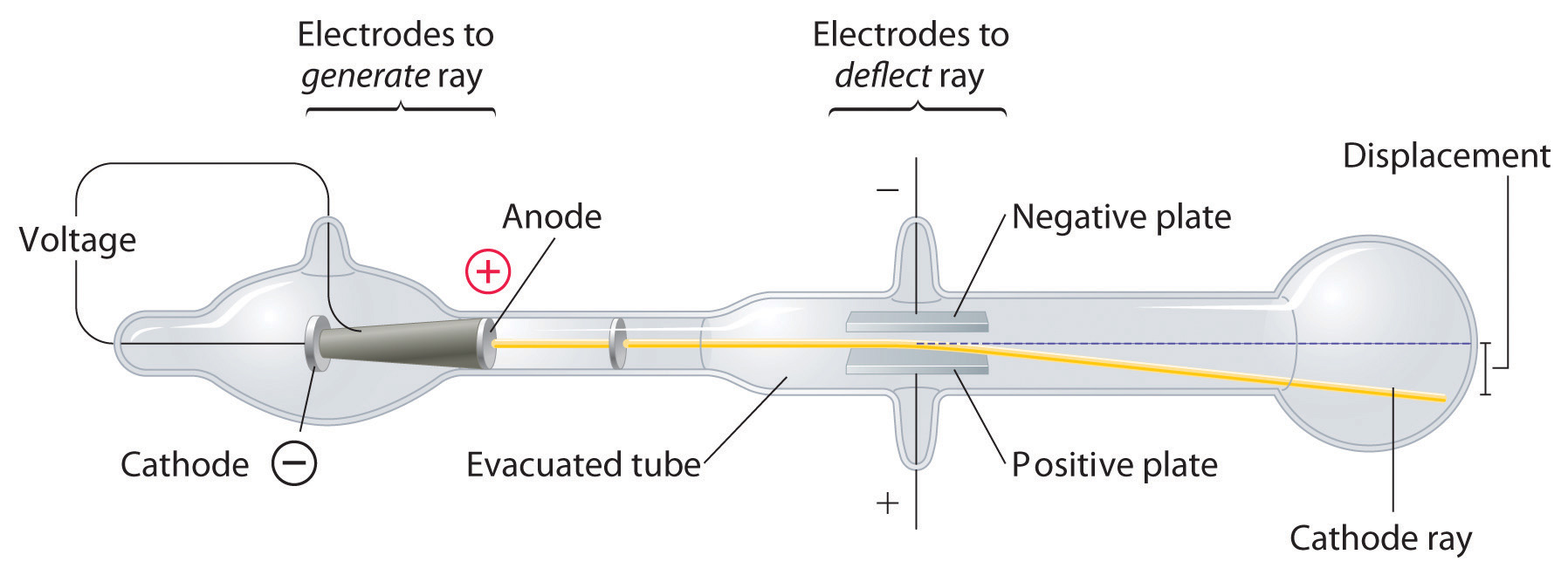
This object consists of a glass cylindrical body with a larger diameter glass ball at one end. Object ID EM.N-08013-B overall length 40 cm, glass ball diam. Thomson's cathode ray tube #3, replica of Cavendish Lab apparatus. Eliot Elisofon Photographic Archives, African Art.In this problem we have given that JJ Thomson cathode ray tube experiment demonstrated that cathode rays are streams of negatively charged ions all the mass of an atom is essential in the nuclear the electron to mass ratio of electron is much greater than electron to mass ratio of proton electron to mass ratio of cathode rays particles changes are different gases placed in the discharge tube to in Thomson experiment there was a discharge due in which in which in which when he died when idiot cathode ray tube experiment to basically cathode rays cathode rays are cathode rays are basically negatively negatively charged rays are stream of cathode rays are streams of negativelyĬharged ions ok negatively charged Ion charged ion and the electron to mass ratio of the electron to mass ratio of electron of electron is basically 1837 time electron to mass ratio charge to mass ratio charge to mass ratio of proton ok so we can say that charge to mass ratio of electron is much higher than the charge to mass ratio of proton because charge of an electron and proton is given as 1.6 into 10 to the power minus 19 coulomb plus minus and mass of electron is given as 9.1 into 10 to the power minus 31 kg and mass of proton is given as 1.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
